Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.
Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.

Regulatory Foundations of Titanium Mesh Labeling and Price Transparency NMPA Medical Device Regulations: Labeling Mandates for Surgical Implants China's National Medical Products Administration (NMPA) classifies titanium mesh as a Class III medical d...
VIEW MOREUnderstanding Regulatory Landscapes for Minimally Invasive Spine DevicesTiered Approval Pathways Across Asia-Pacific, Latin America, and Middle East & AfricaGetting MIS devices into emerging markets means dealing with complex regulatory systems at di...
VIEW MORE
Reducing Soft Tissue Complications with Low-Profile Distal Tibia Medial Plates Hardware Prominence and Its Link to Postoperative Irritation and Revision Surgery The problem of hardware sticking out remains one of the main reasons patients face compl...
VIEW MORE
Clinical Imperative: High Failure Rates Drive Biomechanical Scrutiny Nonunion and hardware failure as red flags in unstable olecranon fractures The complication rates ranging between 15 to 30 percent for unstable olecranon fractures really highlight...
VIEW MOREWhy Real-World Evidence Is Essential for Lumbar Disc ReplacementLimitations of Randomized Trials in Capturing Real-World Practice Complexity RCTs were key to getting lumbar disc replacement approved by regulators back in the day, though nobody ever i...
VIEW MORE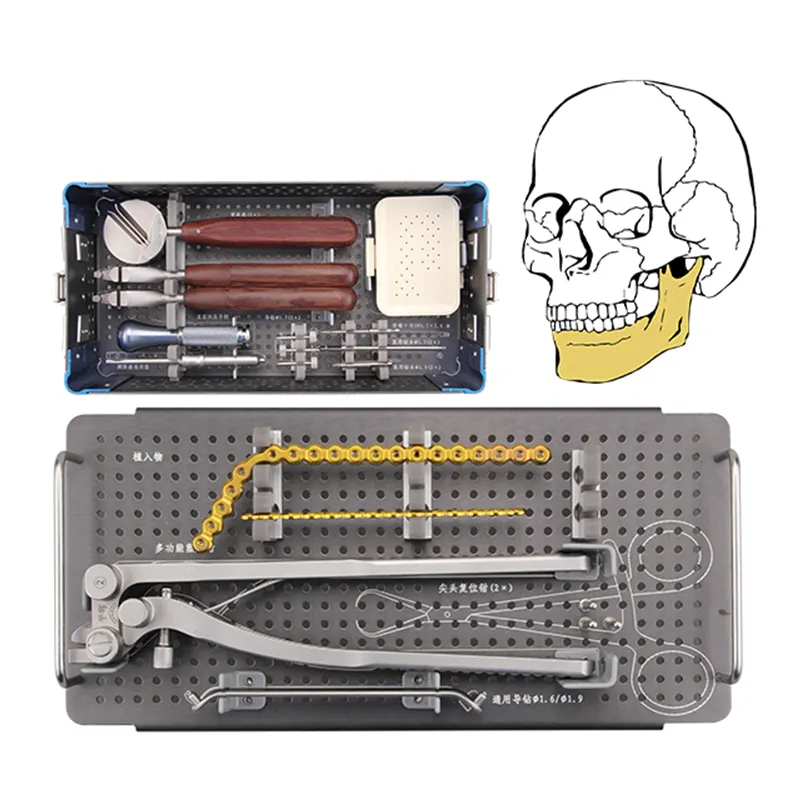
Core Outcome Domains for Maxillofacial Surgery Implant Benchmarking Why Orthopedic Metrics Fail: Anatomy- and Function-Specific Demands of Maxillofacial Surgery Implants The metrics used for orthopedic implants just don't match up with what we see i...
VIEW MORE
ISO 13485 Design Controls Framework for Distal Tibia Locking Plates Implementing ISO 13485 design controls for distal tibia locking plates ensures systematic development of these critical orthopedic devices. The framework mandates rigorous processes ...
VIEW MORE
Regulatory and Clinical Foundations of Bioburden Testing for Sterile Rib PlatesISO 11737-1, USP , and FDA Expectations for Implantable Orthopedic DevicesTesting for bioburden in sterile rib plates follows three main regulations working together: ISO ...
VIEW MORE
Why Design for Manufacturability Matters for Medial Proximal Tibia PlatesThe application of Design for Manufacturability principles completely changes the game when developing orthopedic implants such as those used for medial proximal tibia fractures...
VIEW MORE
How Low-Profile Sternum Titanium Plates Improve Patient ComfortReduced Soft Tissue Irritation Through Anatomically Contoured Design Titanium plates designed for low profile sternum applications significantly reduce tissue irritation because they foll...
VIEW MORE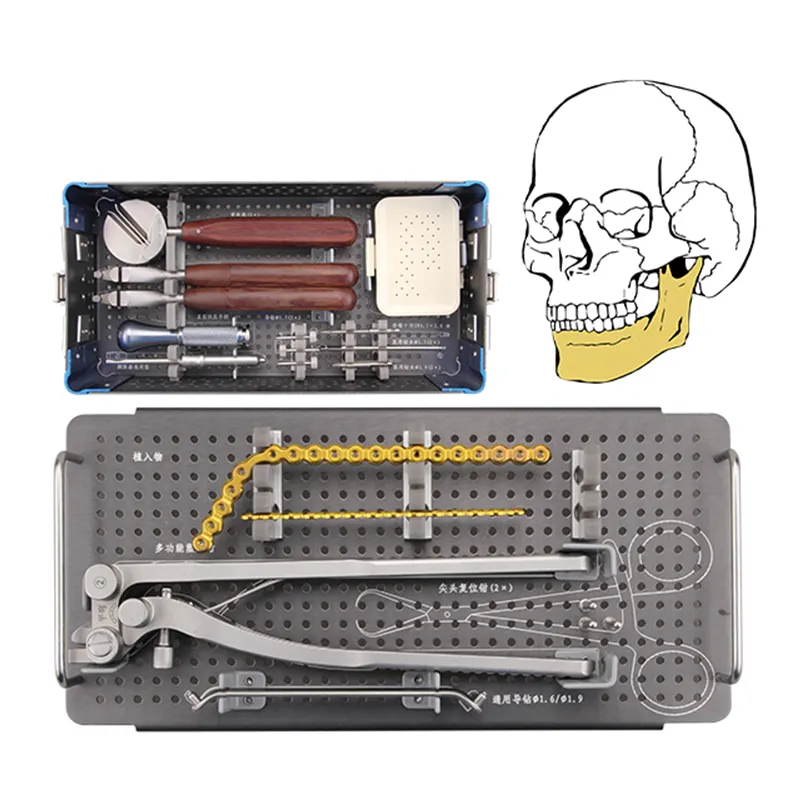
Essential Core Customs Documents for Maxillofacial InstrumentsProper customs documentation for maxillofacial instruments prevents shipment delays and compliance penalties. Accurate paperwork ensures surgical tools clear international borders efficien...
VIEW MORE
Biomechanical Necessity: How Cranial Load Dynamics Demand Sub-0.2 mm Titanium Mesh Stress distribution and strain accommodation in dynamic cranial bone interfaces The bones of the skull experience forces coming from many directions when someone chew...
VIEW MORE