Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.
Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.

Regulatory Foundations and Standards for Shelf Life Validation of Sterile Proximal Femoral Plates FDA Guidance and EU MDR Requirements for Shelf Life Claims on Orthopedic Implants To make legitimate shelf life claims about sterile proximal femoral p...
VIEW MORE
Why Sterilization Validation Is Critical for Heat-Sensitive Absorbable MeshGetting proper sterilization validation right is absolutely essential when dealing with heat sensitive absorbable mesh implants. Just one mistake here could mean serious probl...
VIEW MORE
Why Packaging Integrity Validation Is Critical for Distal Radius Locking Plates The distal radius locking plates need to be completely sterile when they go into patients. If the packaging gets damaged somehow, these orthopedic implants become vulnera...
VIEW MORE
Why Titanium Mesh Composite Demands Specialized Sterilization Methods Key material properties affecting sterilization compatibility The porous structure and mixed makeup of titanium mesh composites present some real problems when it comes to sterili...
VIEW MORE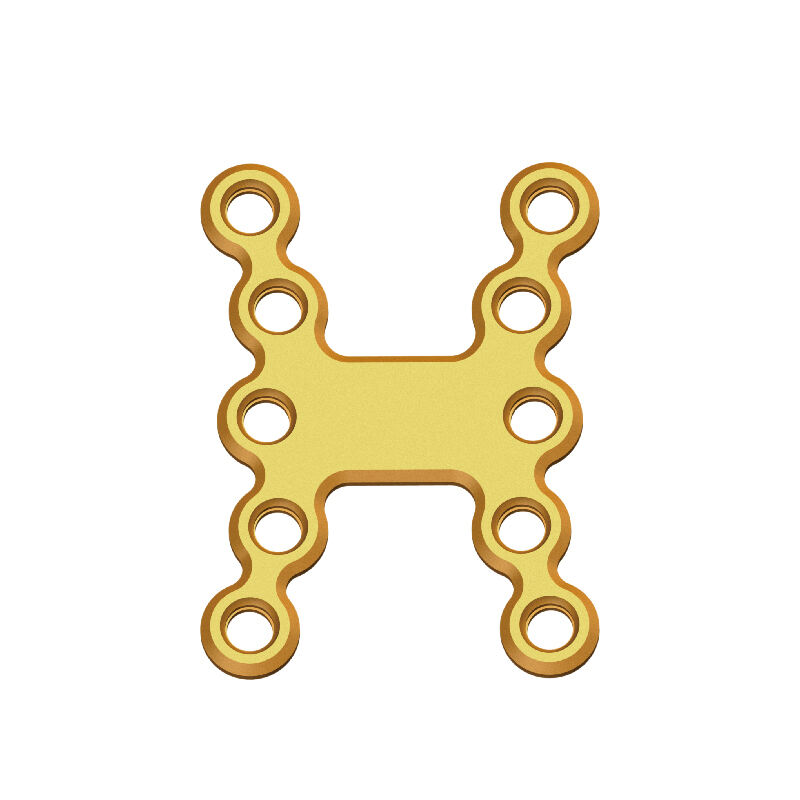
Regulatory Foundations: EU GMP Annex 1 and ISO 14644 AlignmentEU GMP Annex 1 requirements for aseptic processing of sterile orthopedic implantsThe EU GMP Annex 1 sets out pretty strict rules when it comes to keeping things sterile during the producti...
VIEW MORE
Why Predictive Maintenance Is Critical for Rib Plate Surgical TraysHospitals are changing their game when it comes to managing those tricky surgical tray sets with specialized rib plates. Instead of waiting until something breaks or randomly replacin...
VIEW MORE
How Supplier Audit Frequency Directly Affects Trauma Locking Plate Quality Material consistency, dimensional accuracy, and fatigue resistance under audit-deficient conditions When companies don't conduct enough supplier audits, three important quali...
VIEW MORE
Understanding the Cost-Quality Imperative in Distal Humerus Plate ManufacturingFor patients recovering from orthopedic injuries, distal humerus plates need to be strong enough to support healing but not so expensive that hospitals struggle to afford ...
VIEW MORE
The Importance of Cleaning Validation for Orthopedic ImplantsEnsuring patient safety through effective cleaning validation of trauma implantsCleaning validation simply cannot be ignored when it comes to trauma implants such as the Synthes calcaneal p...
VIEW MORE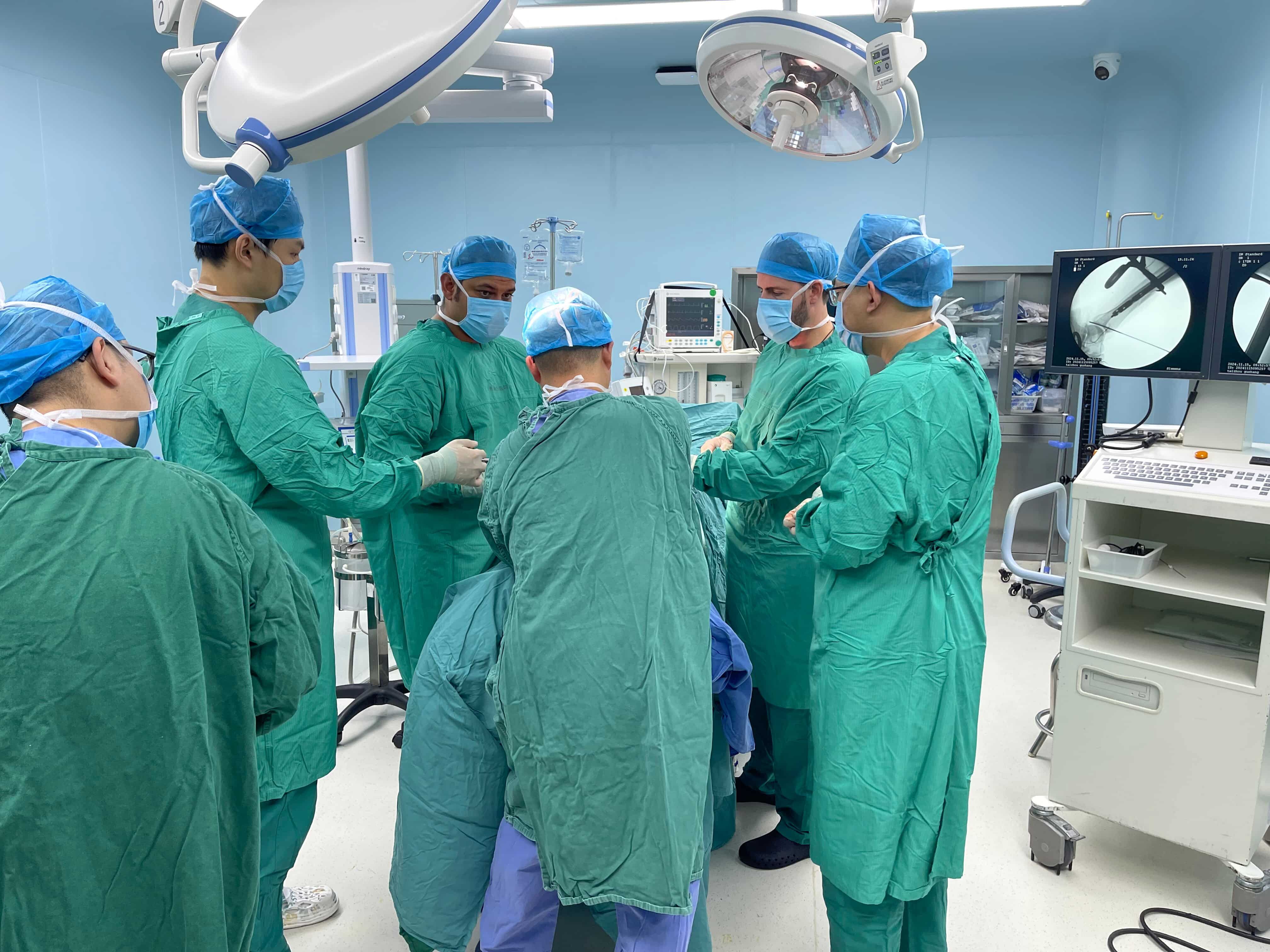
Regulatory Foundations: FDA, AAMI ST79, and ISO 17664 Requirements for Reprocessing Instructions Validation Validating reprocessing instructions for reusable orthopedic instruments is foundational to patient safety and regulatory compliance. Hospital...
VIEW MORE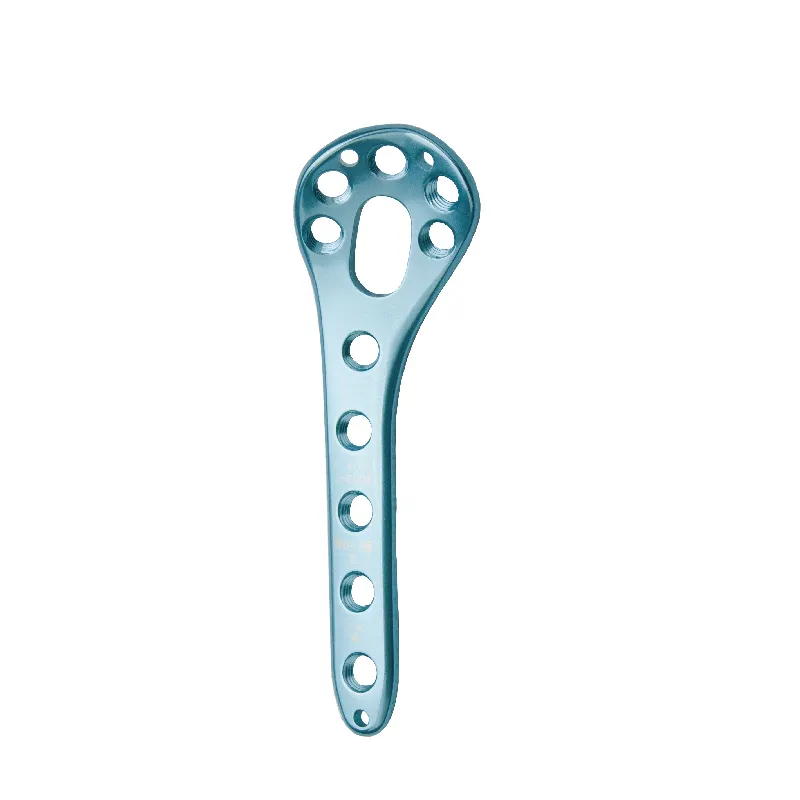
Material-Specific Laser Marking for Optimal Durability Laser Annealing vs. Etching on Titanium and Stainless Steel Laser annealing produces long lasting, high contrast markings on medical grade titanium and stainless steel by creating a permanent ox...
VIEW MORE
Concentrated Global Production: The Core of Orthopedic Implant Supply Chain Risk Orthopedic implant makers face a serious problem because most of the world's ASTM F136 compliant titanium sponge comes from just a few places. This titanium sponge is b...
VIEW MORE