Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.
Shuangyang Building, Yangshe Town, Zhangjiagang City, Jiangsu Province, China.

Standardizing Core Manufacturing Processes for Multi-Site ProductionMaterial Specifications, Heat Treatment, and Surface Finish HarmonizationGetting consistent manufacturing results across different production sites starts with making sure all the ma...
VIEW MORE
Ensuring Patient Safety and Material Biocompatibility Through ISO 13485 ISO 13485 certification for medical device material suppliers: A foundation for patient safety The ISO 13485 standard creates what many call a risk-focused quality system that's ...
VIEW MORE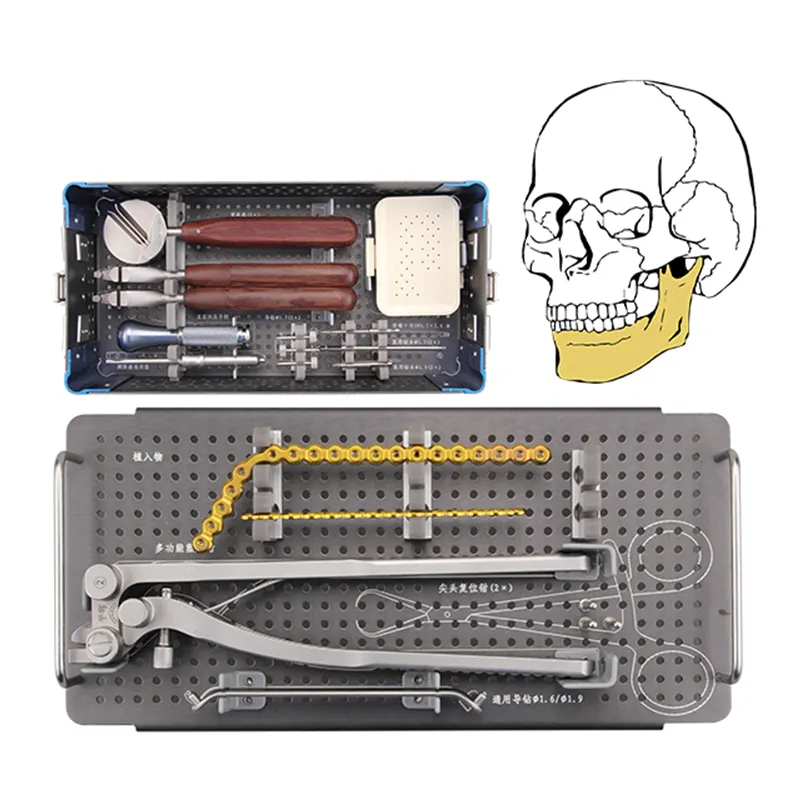
Why QMS Software Is Essential for Maxillofacial Prosthodontics ComplianceThe regulatory stakes: ISO 13485, FDA 21 CFR Part 820, and EU MDR in craniofacial device manufacturingManufacturers of craniofacial devices need to follow several key standards ...
VIEW MORE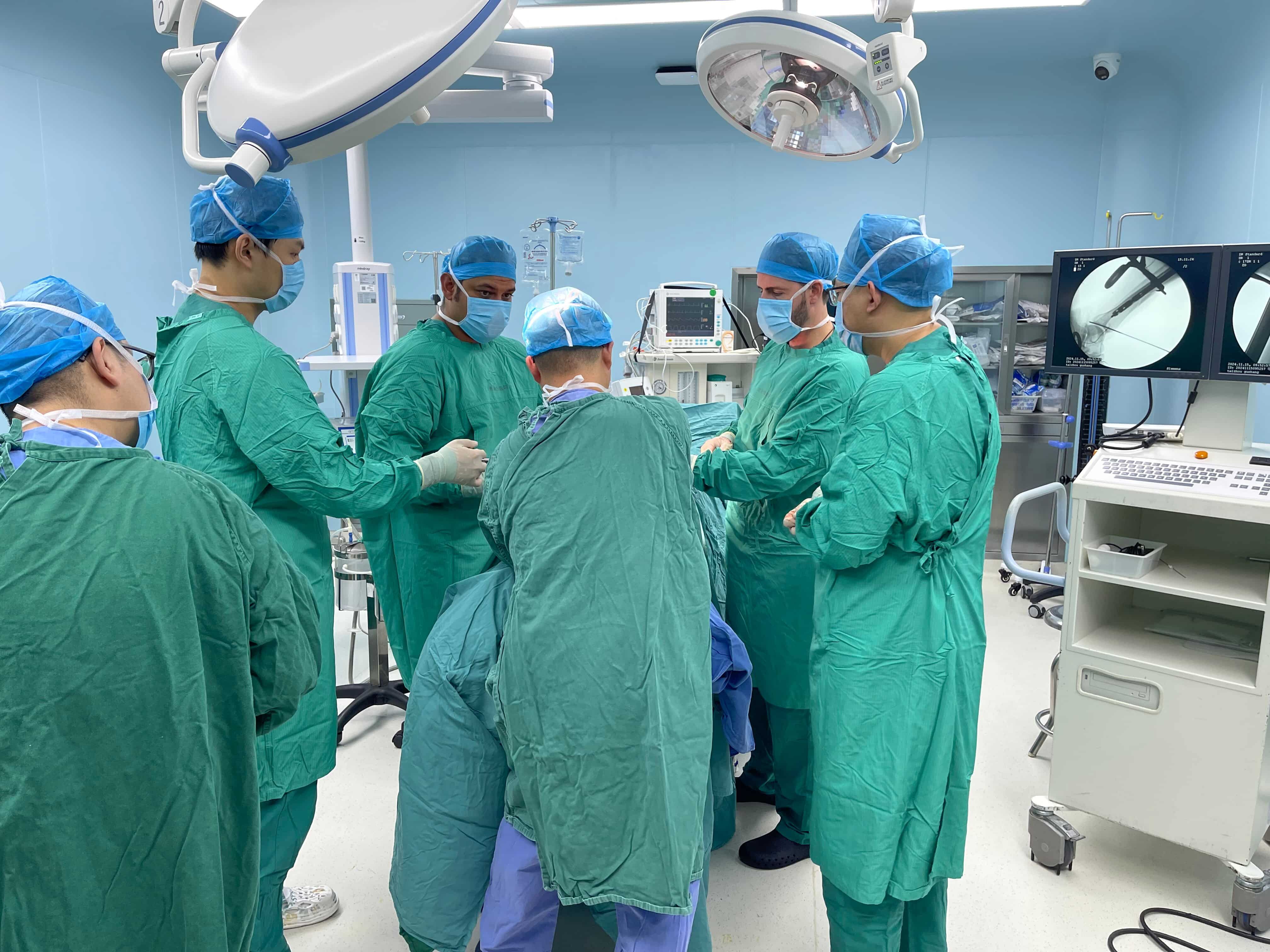
Understanding the Threat of Counterfeit Titanium Cables in Mission-Critical Industries Prevalence of counterfeit parts in aerospace and defense supply chains Fake parts keep getting into aerospace and defense supply chains, especially titanium cable...
VIEW MORE
Why Olecranon Plate Geometry Challenges Traditional Cleaning ValidationTopographic Complexity: Undercuts, Porous Surfaces, and Additive Manufacturing ArtifactsThe complex shapes of olecranon plates make them particularly difficult to clean properly. ...
VIEW MORE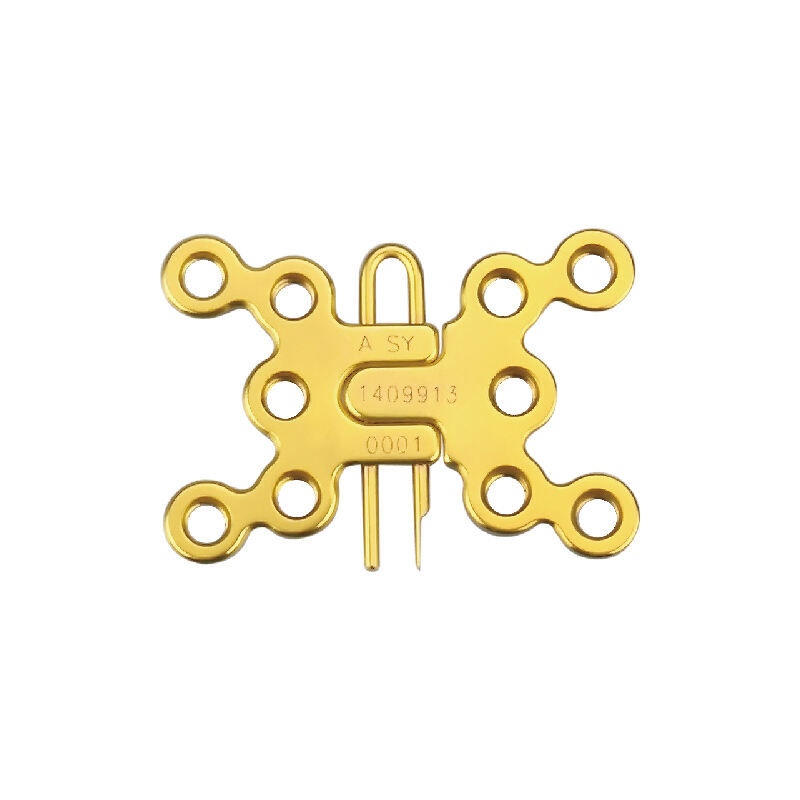
What Is Lot Traceability in Medical Devices?Definition and Core Concepts of Lot TraceabilityTracking medical devices through their production lots means following items made from the same materials using similar processes at around the same time. Eve...
VIEW MORE
Assess Demand Drivers for Distal Femoral Fracture Plates Rising Incidence and Seasonal Trauma Patterns Impacting Plate Utilization The number of distal femoral fractures definitely goes up and down with the seasons. Winter tends to bring about 20 to...
VIEW MORE
Why Component Obsolescence Threatens Long-Term Serviceability of Proximal Tibia Plates The Lifecycle Mismatch: Medical Device Longevity vs. Electronics & Material Supply Chains Most proximal tibia locking plates should last patients well over two de...
VIEW MORE
Regulatory Foundations of Supplier Qualification for Implant Screws FDA 21 CFR 820.50 and EU MDR Annex II Requirements for Critical Component Suppliers The FDA regulation 21 CFR 820.50 puts strict rules on how medical device makers must qualify thei...
VIEW MORE
The Science Behind Anodization Color Formation in Medical-Grade Aluminum Why anodization color consistency matters in orthopedic surgery tools During complicated orthopedic surgeries, doctors depend heavily on what they see when working with tools. ...
VIEW MORE
Thermal Behavior of Titanium Alloys During Common Sterilization Processes Phase stability and microstrain accumulation in CP-Ti and Ti-6Al-4V under autoclave (134°C) vs. dry heat (160–180°C) conditions Titanium alloys CP-Ti and Ti-6Al...
VIEW MORE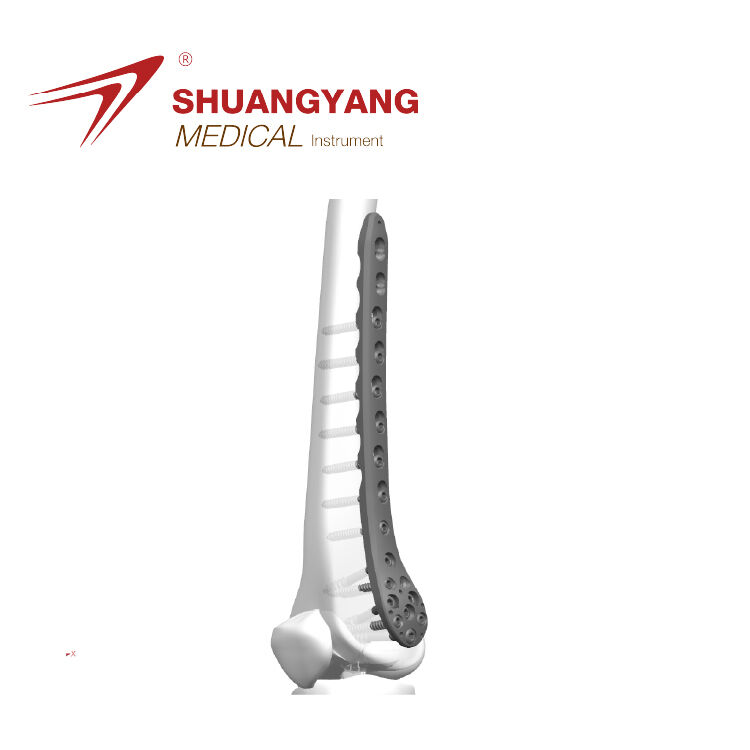
Applying Core Lean Manufacturing Principles to Orthopedic Implant Assembly Mapping the Value Stream in Distal Humerus Plate Production Value stream mapping, or VSM for short, helps manufacturers find places where they can cut down on waste during th...
VIEW MORE